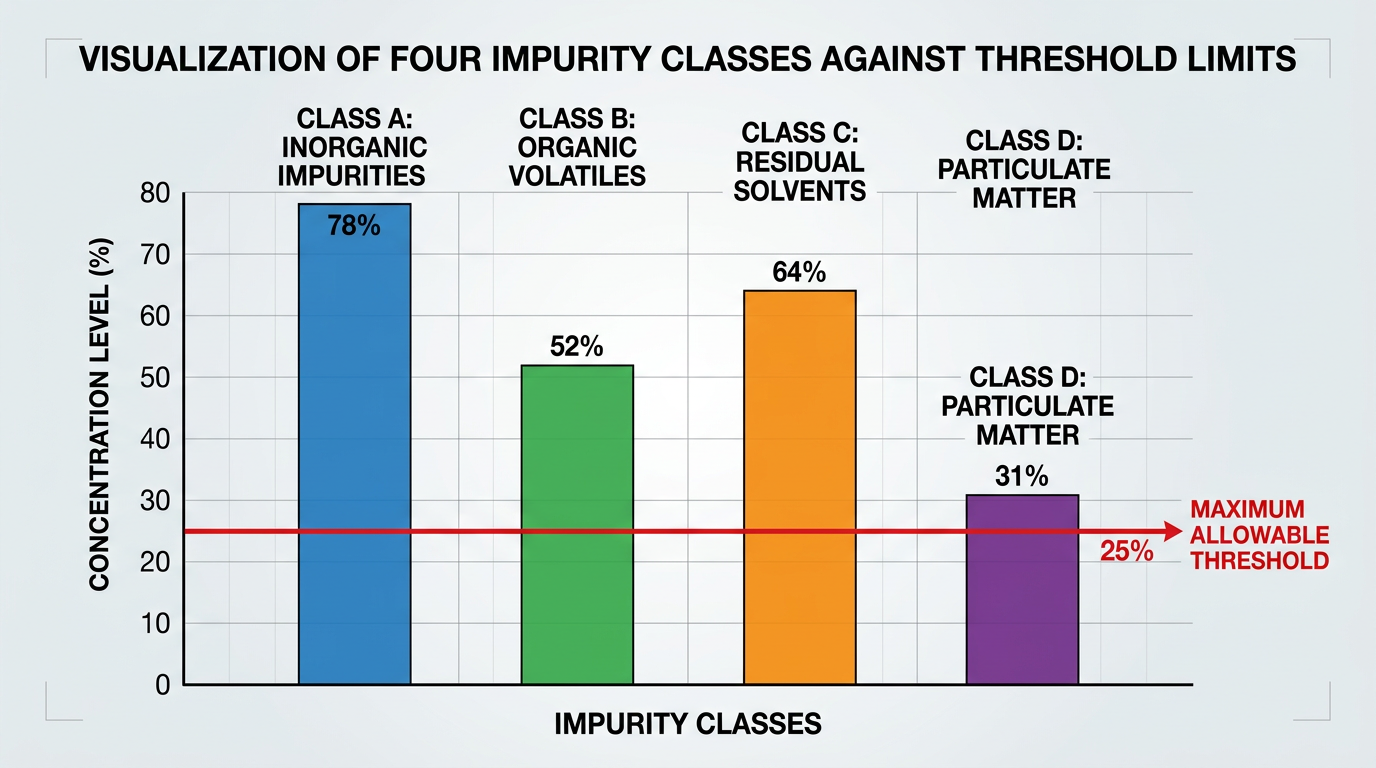
In 2006, the EU Scientific Committee on Consumer Products tested ten production batches of hydroxybenzomorpholine. Total nitrosamine ran 492 to 2616 ppb. That is 10 to 50 times over the 50 ppb cap, which has since hardened into the SCCS 50 µg/kg ceiling under Regulation 1223/2009.
The route a CMO actually runs therefore matters as much as the COA purity number. The published commercial route for hydroxybenzomorpholine (CAS 26021-57-8) is the four-step telescoping synthesis from Wella Germany Gmbh’s WO2025099111A1 (priority November 2023). The four steps below — ring closure, nitro reduction, 6-OH installation, lactam reduction — are walked through in order.
Step 1 — Ring Closure to the Morpholinone
The first step builds the morpholinone (the lactam scaffold) from 2-amino-4-nitrophenol. Chloroacetyl chloride acylates the aniline nitrogen in K2CO3/DMF. The ring then closes to give 6-nitro-4H-1,4-benzoxazin-3-one in 85% yield.
On a kilo scale this runs in a jacketed reactor with controlled chloroacetyl-chloride dosing. The acylation is exothermic. Dosing rate sets both the impurity profile and the temperature excursion.
The impurity that carries forward to the COA is residual chloroacetate. It is genotoxic and has to be specced at ppm. A defensible RFQ specifies chloroacetate hydrolysis products at trace level, with residual aminophenol below 0.1% by HPLC area as a reasonable target.
What the buyer is checking is whether the cyclization clean-up actually drove chloroacetate down. Aqueous wash plus crystallization is the standard polish. Without that polish, the supplier ships kinetic product and the COA’s chloroacetate line will reveal it.
Step 2 — Catalytic Reduction of the Nitro Group
The second step reduces the aromatic nitro group to the corresponding amine. Pd/C catalytic hydrogenation runs in ethanol at 3 bar H2. The published conditions are 10 wt% Pd/C 50%-water-wet at 5 mol% loading, delivering 6-amino-4H-1,4-benzoxazin-3-one in 64% yield.
LiAlH4 is the reductant a CMO walks away from for an aromatic-nitro step. Fire risk, off-gas burden, and aqueous quench dominate the cost stack on anything above lab grams. Pd/C wins because it tolerates moisture, survives acidic and basic conditions, and recycles through filtration with limited activity loss.
The dominant control problem is palladium leaching off the carbon support, which lands directly on the COA’s metals line. The Pd<1 ppm target the SCCP reported for batch 0508918 is the implied ceiling, not a stretch goal. A kg-scale run at 5 mol% Pd loading without a deliberate Pd-scavenging polish will not hit it.
The buyer-side check is that the COA reports Pd by ICP-MS as a discrete line. Bundling into “≤10 ppm total heavy metals” lets aggregate metals hide an out-of-spec Pd number.

Step 3 — Aromatic Hydroxylation at the 6-Position
The third step installs the 6-OH on the aromatic ring through diazotization-hydrolysis. The 6-amino lactam from Step 2 is diazotized with NaNO2 in dilute H2SO4 below 5 °C. Acid hydrolysis of the diazonium salt then gives the 6-hydroxy lactam (Example 3 of WO2025099111A1).
Diazonium chemistry on this substrate is sensitive to nitrite-stream control. Residual nitrite in contact with the secondary amine can regenerate N-nitroso byproducts. That is the chemistry that historically produced NDELA and NBHPA traces in cosmetic products and the chemistry the SCCS 50 µg/kg ceiling exists to prevent.
A second route to the 6-OH installation uses Cu-catalyzed alkaline hydrolysis of a halogenated precursor. Conditions are KOH / CuI / diethylene glycol dimethyl ether, 215 °C, 8 h, in a steel autoclave. The 6-bromo precursor delivers 60% to HBM, while the 6-chloro analogue gives only 3%.
A 20× yield gap between bromo and chloro is the kind of due-diligence question to put on a CMO RFQ. A supplier quoting the chloride route is admitting either a different precursor strategy or a 20× yield penalty on this step.
Step 4 — Reduction of the Lactam to the Morpholine
The final step reduces the lactam carbonyl to the corresponding morpholine. Borane-dimethylsulfide complex in THF at reflux is the kg-scale reductant. The published conditions use 3.75 equivalents over roughly 2 hours, giving 3,4-dihydro-2H-1,4-benzoxazin-6-ol — HBM itself — in 75% yield.
Borane-Me2S is chosen over LiAlH4 because the aqueous quench is manageable. Acidic workup decomposes the borane-amine adduct cleanly. LiAlH4 quench at lactam-reduction scale is one of the harder unit operations to engineer safely.
The remaining moves are crystallization, filtration, and drying to cosmetic-grade specification. The final purification step is also where the COA panels are run.
The COA needs four panels reported as discrete lines, not a single “≥99% pure” headline:
- ICP-MS for the metals line (Pd from Step 2, Ni and Fe from reactor wear)
- GC for residual solvents (DMF carry-forward from Step 1, THF from Step 4)
- HPLC for purity and residual starting materials
- LC-MS/MS for total N-nitrosamines and free secondary amine per SCCS/1458/11 (≤50 µg/kg total nitrosamines, ≤0.5% free secondary-amine impurity in the raw material)
The hydroxybenzomorpholine (CAS 26021-57-8) spec sheet is the kind of document to bring to a CMO RFQ when those four classes get translated into release criteria.

Putting the Four Steps Together
The four steps add up to the molecule by walking the substrate through three transformations on the aromatic core (ring closure, nitro reduction, 6-OH installation) plus one transformation on the heterocycle (lactam reduction). Each step’s overall yield (85% / 64% / Example-3 / 75%) compounds to roughly 30-40% from 2-amino-4-nitrophenol.
A single-pot variant exists. Zhejiang Dinglong’s CN111170958A (granted 2021) telescopes demethylation and ring closure in water using a recyclable solid-acid catalyst — SO4²⁻/ZrO2-MnO2 at 110 °C for 4 h, with 79.8% yield in Example 4. It is a one-pot replacement for the cyclization stage, not a parallel four-step process.
Each step leaves a different fingerprint on the COA. The four classes are chloroacetate from Step 1, Pd from Step 2, N-nitroso risk from Step 3, and borane residue plus the final-purification panel from Step 4. An RFQ that doesn’t separate these into discrete lines lets the supplier ship a chemically-pure but cosmetically-non-compliant batch — which is what the SCCP’s 2006 survey found at 492-2616 ppb against 98-99% HPLC purity.